
Press release
WBVR and CEPI announce collaboration to evaluate vaccines against the COVID-19 coronavirus
Wageningen Bioveterinary Research (WBVR) and the Coalition for Epidemic Preparedness Innovations (CEPI) have started a new partnership to facilitate vaccine development against SARS coronavirus 2 (SARS-CoV-2), the virus that causes COVID-19. In this partnership, WBVR will develop preclinical models for SARS-CoV-2, so that vaccines against the virus can be tested for efficacy and safety in humans.
Developing vaccines and antivirals
SARS-CoV-2 first emerged in the Wuhan region of China in December 2019 and is rapidly spreading worldwide. Scientists across the globe are busy developing vaccines and antivirals to prevent or treat the disease.
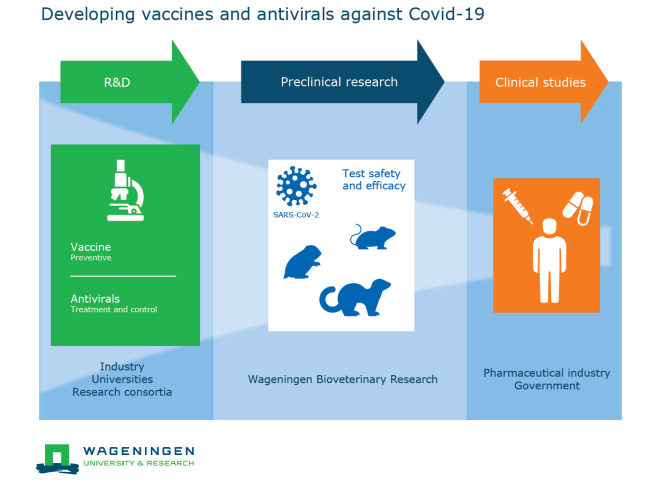
Testing for safety and efficacy
CEPI coordinates and funds the worldwide development of vaccines in the fight against COVID-19. In this collaborative project, WBVR will develop preclinical models for COVID-19, which are indispensable for the development of safe and effective antivirals and vaccines needed to finally control the coronavirus outbreak.
Facilities of WBVR
Ludo Hellebrekers, director Wageningen Bioveterinary Research: “Through this partnership, CEPI is making it possible for WBVR to make a substantial contribution to public health. With the animal models being developed, we will be able to test vaccines that can be used for the control and prevention of SARS-CoV-2, in the specialized facilities of WBVR”.