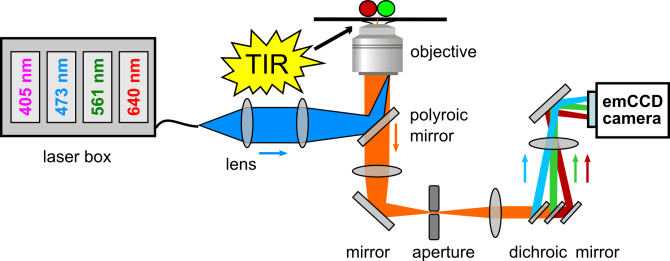
Equipment
Single-molecule Förster Resonance Energy Transfer (smFRET)
FRET is used to measure molecular scale distances (1-10 nm) between fluorescence tags in individual proteins / protein complexes / protein-DNA complexes. FRET is measured on a single molecule level with a total internal reflection (TIRF) microscope with 4 (alternating) laser excitation wavelengths and three wide field emission channels.
Single-molecule Förster Resonance Energy Transfer is a well-established technique to measure distances between two fluorophores with high spatial and temporal resolution. Theodor Förster showed in 1948 that the energy from an excited donor fluorophore can be transferred to an acceptor fluorophore as long as the emission spectrum of the donor overlaps with the absorption spectrum of the acceptor and both fluorophores are in close proximity (<10 nm). The proportion of transferred energy E depends strongly on the distance r between the two fluorophores (E is proportional 1/r^6) making the technique useful to study, for example, conformational changes and dynamics of enzymes such as DNA polymerases.
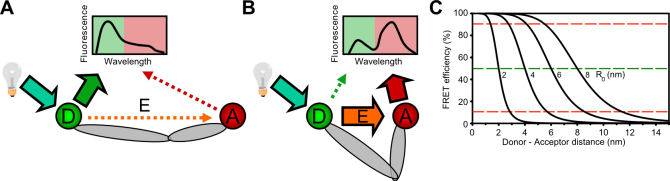
(A) A donor fluorophore (green) is excited by a light source and attached to a flexible biomolecule. As long as the acceptor dye (red) is far away (>10 nm) from the donor, the emission spectrum of the donor is not significantly altered (zero or low FRET).
(B) If the distance between the fluorophores decreases, more energy is transferred from the donor to the acceptor and the entire emission spectrum shows a significant contribution of the acceptor flurophore emitting lower energy photons.
(C) FRET efficiency plotted as a function of the distance between the fluorophores for various Förster radi (R0). The Förster radius can be calculated for specific dye pairs and is normally between 5 and 7 nm.