
News
Efficient selenium recovery from waste streams
Selenium is an important element used in a variety of industrial applications, such as photo-electric cells and solar cells because it can convert light into electricity. In addition, selenium is a trace element necessary to maintain good health in humans and animals. However, current resources of this valuable element are limited and possibly exhausted in less than 50 years, urging its recovery and recycling.
In contrast to its valuable health properties when consumed in small quantities, selenium is potentially toxic in larger amount and release into the environment should be avoided. ‘Several waste streams from the industry contain selenium’, Hageman says. ‘It is among others present in the emission gasses from coal plants. When these gasses are washed before release, selenium ends up in the wastewater. Therefore, technology is needed to recover this element.’ Selenium recovery from wastewater not only prevents environmental pollution. At the same time, these waste streams may function as a much needed selenium resource.
Selective method
Removal of selenium dissolved in wastewater is no easy task. As a first step, this element needs to be converted in a solid form. This makes recovery a lot easier. Transforming dissolved selenium into solid selenium is possible using strong chemicals, but a drawback is that this method is not selective: many compounds present in wastewater precipitate resulting in a mixture of many different compounds. A more subtle and selective method is by using microorganisms. ‘Wastewater contains dissolved selenium in two main oxidized forms: selenate and selenite’, Hageman explains. ‘Microorganisms can convert the first form, selenate, directly into selenium. This consequently precipitates in amorphous, nano-scale sized particles.’ Due to their size, these particles are quite difficult to recover. Hageman discovered that by manipulating reaction conditions like temperature and pH, the size of precipitated selenium particles could be influenced. By increasing the temperature to 50 °C and keeping high pH, around 8 or 9, relatively large selenium crystals were formed, Hageman found out. Hageman: ‘Selenium particle formation can clearly be controlled. These larger, crystalline selenium particles can be recovered more easily and makes the separation economically feasible.’
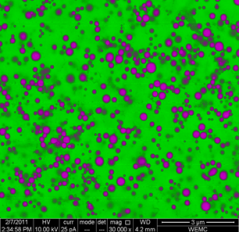
Large selenium particles
During the experiments, Hageman found an even better process for selenium recovery. Selenate is very stable and not easy to transform. The other form however, selenite, is highly reactive and may more easily be converted into selenium. The scientists managed to find out the specific reactor conditions where almost all selenate was transformed into selenite: that occurred at a temperature of 30 °C and neutral pH.

Sulphide, formed by microorganisms in waste streams, can react with selenite to form seleniumsulphide that precipitates. Microorganisms present in sludge subsequently convert seleniumsulphide back into sulphide and solid selenium. The selenium thus recovered had a crystalline structure, while particles were visible and relatively large, in the order of micrometers.
Proof of principle
According to Hageman, this indirect route, via selenite, to recover selenium, is more promising than the direct microbiological conversion from selenate into selenium. The required temperature is lower, saving energy, while sulphide is continuously recycled during the reaction process. In addition, the method is practical and easily applied, while the recovered selenium particles are larger, enabling a more efficient recovery. ‘The proof of principle is there, the final step will be to test the method in a practical industrial setting’, says Hageman.