
News
Science: Combining different technologies for cleaningsaline wastewater with organic contamination: a step-by-step solution to the problem
Together with project partner Dow Benelux in Terneuzen, ETE researcher Pradip Saha developed a method to clean saline, industrial wastewater. A complex task, since single technologies didn’t do the job. Therefore, he combined three different technologies for an effective cleaning: oxidation to remove most organic compounds followed by the use of constructed wetlands to remove some remaining organics. The last step included salt removal by standard membrane desalination technology.
During the manufacturing of chemicals, company Dow Benelux uses substantial amounts of fresh water in their production process. A significant part of their yearly 22 million m3 fresh water consumption is used for cooling. But in the process this cooling water becomes contaminated. Corrosion and scale inhibitors and biocides are added to prevent scaling and excessive bacterial growth. In addition, due to evaporation, salt concentration increases. The end result is the so-called cooling tower blow-down water, that contains a complex mixture of salts and organic compounds. Yearly, around 1 million m3 of this cooling water is discharged. ‘We aim to reduce our fresh water use’, Niels Groot (see also column), Water Technology professor at HZ University of Applied
Sciences and a water specialist at Dow Benelux, says. ‘We don’t want to compete for scarce resources.’ Therefore, for a more sustainable operation, Dow aims to reduce the use of fresh water by cleaning and reusing the cooling water.
Challenging
However, removing this cocktail of salt and organic substances is
challenging. Salt can be removed by by existing membrane technologies, but the organic compounds tend to clog the membranes’, Saha says. ‘Therefore, we need to remove the organics before desalination.’ Since the presence of salt limits microbial degradation of these organic components, Saha had to explore other methods. Previously, Dow explored the possibility to coagulate and subsequently filtrate the wastewater to remove the organics, but this method proved not effective enough.
The application of electrochemical oxidation (EO) was more successful. The principle of this technology is to generate radicals from water by electricity. These radicals consequently degrade the organic substances present. ‘By applying current over a cathode and a specially diamond-coated anode, water can be split into a proton (H+) and a hydroxyl radical (OH.)’, Saha explains. ‘This radical actively degrades the organics, resulting in a 60-70 percent removal. This likely is enough to prevent clogging of the membranes used in the desalination process.’
Major problem
But the successful method proved to have a major problem: the formation of toxic compounds. EO converted the chloride (Cl-) present in wastewater into the reactive chlorine species. That compound consequently reacted with certain organic substances into highly toxic halide compounds. A
first attempt to remove the toxic halide by constructed wetlands was
successful, but eventually the plants and bacteria died because halide
concentrations were too high. Therefore, the method could not be used and an alternative method had to be found. The scientist went back to the drawing table to find a technology to produce radicals in a completely different way, that avoided the formation of toxic compounds.
Promising
A better option was found in the persulfate oxidation method, a technology that produces sulfate radicals without the formation of toxic compounds. ‘By simply adding heat to persulfate, a sulfate radical is formed’, Saha explains. ‘The heat needed can be extracted from the process, so no additional energy is needed.’ The results from Saha’s
experiments look promising: almost all organics were removed, without the formation of toxic chlorinated by-products. There is however formation of some sulfate in the process.

To add an additional cleaning step where constructed wetlands remove these sulfates, as well as some nitrates, will improve the overall cleaning before desalination.
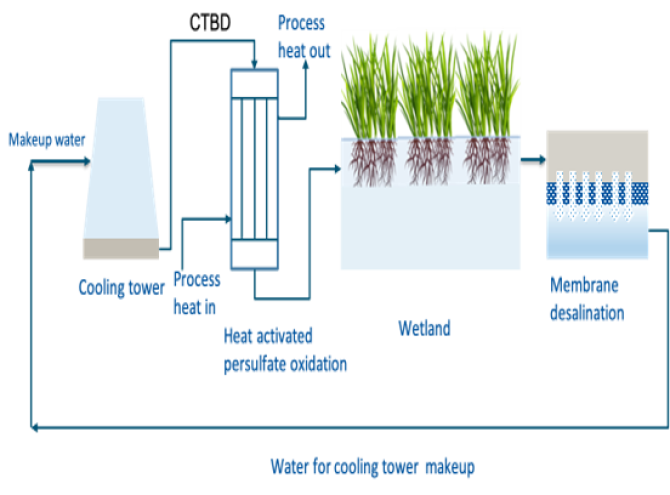
Fig 2. Integrated technology train for cooling tower blowdown (CTBD) treatment
Huge advantage
Despite the success of using sulfate radicals to remove organics, the scientist is now experimenting with UV light as an alternative method to degrade the organic substances. ‘For the persulfate oxidation, you need to add sulfate’, Pradip says. ‘Ideally, you don’t want to add any more chemicals.’ The first results for the UV oxidation experiments reveal significant organic compound removal, enough to proceed with
desalination. However, this method requires the input of extra energy. Project partner Dow really appreciates that the ETE’s research has resulted in two attractive options for reusing fresh water, but they have a clear preference. ‘In our production, there are quite a few waste streams containing sulfate, and these could be used as a resource for the persulfate oxidation method’, Groot says. ‘Together with using heat from the process, this is a huge advantage since we can reuse both sulfate waste and heat.’
Publication
Saha, P., Wagner, T.V., Ni, J., Langenhoff, A.A.M., Harry Bruning, H. & Rijnaarts H.H.M. 2020. Cooling tower water treatment using a combination
of electrochemical oxidation and constructed wetlands. Process Safety and
Environmental Protection 144: 42-51.