
News
Science: Better metal recovery for improved yield and less mining waste
ETE scientist Adrian Hidalgo-Ulloa developed a new method to improve the retrieval of metals from mining streams. He redesigned the recovery process of commercial mining companies: by adding hydrogen sulfide (H2S) to the mined metal-containing rocks, crystallized metal salts were formed. By manipulating the pH, he was able to precipitate the different metal salts one by one making an easy recovery possible. Hidalgo-Ulloa: ‘With this method we have potentially increased the metal recovery yield, but also reduced the metal waste in mining tailings tremendously.’
Due to the increased electrification and the transition to a more electricity-based society, the demand for metals, like copper, nickel, zinc and cobalt, is growing at a high rate. For example, global copper demand is estimated to double to 50 million tons by 2035, while prices are expected to increase as well. This puts extra pressure on current mining companies to expand their activities, to meet the growing needs. However, the environmental impact of these activities will increase as well. ‘One of the issues is that the currently low metal prices do not reflect the environmental costs of mining’, Hidalgo-Ulloa says. ‘There is no incentive to be efficient with the recovery of metals. Often it is cheaper to waste metals, than to put money and effort into a more efficient operation.’
Low efficiency
Mining operations are usually quite straightforward: metal-containing rocks are mined, and grinded to small particles for an easier metal recovery. Then, acids and water are added and the mixture is heated to dissolve the metals. Adding caustic soda (NaOH) will increase the pH, resulting in the precipitation of the target metals as salt crystals, for example copper-hydroxide, that can be recovered. Since mining companies often target only a few metals, while the mined rock often contains many different metals, there is a low efficiency of metal recovery: as much as 40-60 percent can be wasted. These waste streams, ‘mine tailings’ are stored on land and obviously pose an environmental hazard due to leaching of toxic compounds in to the soil and ground water. Hidalgo-Ulloa: ‘In 2020, the total amount of mining waste produced equaled more than 216 cubic kilometers, just because of inefficient metal recovery.’
Highly toxic compound
During his PhD research, Hidalgo-Ulloa developed a better method for metal recovery: he used hydrogen sulfide (H2S) to react with the metals, forming metal-sulfides. The advantage of these salts is that their solubility changes with changing pH, allowing for an efficient recovery of most metals present. ‘At acidic pHs, most metal sulfides are soluble. Increasing the pH affects the solubility of most metal sulfides greatly, but individually. So, when one metal sulfide is almost insoluble, the others remain partially in solution. For example, at pH 3, copper sulfide is insoluble, while nickel-sulfide is quite soluble’, Hidalgo-Ulloa explains. ‘By producing H2S into the same vessel as the acid rock mixture and slowly increasing pH step by step, different metal salts precipitate, permitting their one-by-one recovery.’ However, to make this work in a cost-effective way, the H2S production and transport should be profitable. Ideally, transporting this highly toxic compound to the recovery plant should be at low cost, or avoided altogether. In the Dominican Repu-blic, the Pueblo Viejo Mine developed a reactor design for the metal sulfide precipitation, with a separate compartment for H2S generation on site (Fig. 1).
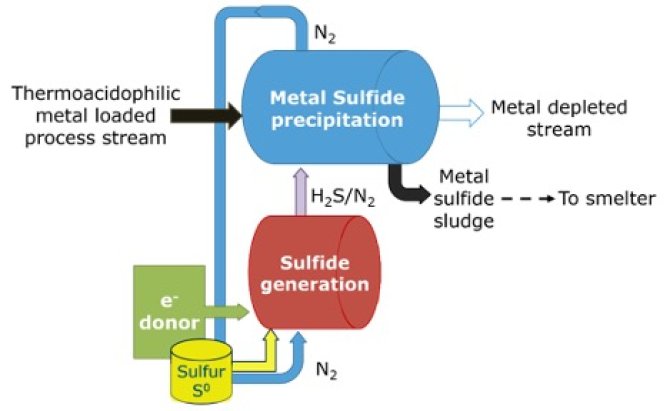
However, it would be even more cost-efficient to design a reactor where the H2S formation and metal sulfide precipitation occurred in the same vessel. The H2S formed could then directly react with the metals, forming metal sulfides that could subsequently be precipitated (Fig. 2).

Test reactor
For the first challenge, to make H2S at low cost, Hidalgo-Ulloa used
microorganisms to do the job. There are two possible methods to do this: the
first one uses sulfate (SO42-) and hydrogen that react to
H2S, while the second method uses elemental sulfur and hydrogen.
‘The second method, based on elemental sulfur, is four times more efficient
than the sulfate method, so we used this one to be as efficient as possible’,
Hidalgo-Ulloa explains. ‘Our next focus was on making this work in a single
reactor vessel,where all reactions would preferably take place in one single tank.’ But this approach was challenging, because the reactor conditions with low pH, high temperature and toxic metals present are highly unfavorable for most microorganisms.
Suitable microbes
Therefore, as a first step, the scientists aimed to find suitable microbes that were up to the task. They focused on industrial reactors to find suitable microorganisms. So, they collected microorganism samples from different industrial reactors and tested their activity and H2S production in a gas-lift reactor with a low pH, between 2 and 4 and high temperature, up to 80 °C. Eventually, Hidalgo-Ulloa found a suitable culture that originated from a reactor from a chemical plant in Emmen: the microorganisms survived the reactor conditions while producing H2S. ‘We were lucky to find those microorganisms’, he says. In addition, the culture tolerated metals really well, and eventually we found the conditions to increase their activity and H2S production! Further optimization, with lower temperature and increasing the amount of metals, resulted in a 10-fold increase in H2S production. Nevertheless, despite the massive improvements, the H2S yield is still on the lower limit for industrial purposes. ‘There are still a lot of optimizations possible and rates might be pushed to add even more metals and fine tune parameters like temperature and reactor flow rate’, Hidalgo-Ulloa says. ‘The main issue however remains that the price of the environmental damage should be included in mining operations, so mining companies have a stronger incentive to be more efficient and less polluting.’

Selected publication:
Hidalgo-Ulloa A., Buisman C.J.N., and Weijma J. 2022. Metal sulfide precipitation mediated by an elemental sulfur-reducing thermoacidophilic microbial culture from a full-scale anaerobic reactor. Hydrometallurgy 213, 105950