
Precision nutrition in cardiometabolic health
We are dealing with a worldwide increase in the development of overweight-related chronic metabolic diseases. An effective way to prevent diseases such as type 2 Diabetes mellitus (T2D) and cardiovascular diseases is via dietary and lifestyle interventions. Although nutritionists have long been aware that what works for one person may not work for another, nutritional advice is still given at a population level via general nutritional guidelines reliant on the group mean (one-size-fits-all).
To improve the effectiveness of lifestyle intervention, it is important to move beyond current public health dietary recommendations and apply a personalized dietary approach. To identify variations in response to diet and explain the underlying mechanisms, we performed several state-of-the-art dietary intervention studies. In these studies, the effects of nutrition on health were magnified by comprehensively phenotyping a person’s metabolic health state by not only performing measurements in the fasted state but also after a meal challenge test. By combining cutting-edge omics technologies, such as transcriptomics and metabolomics, we were able to identify unexplored mechanisms of diet on metabolic health. For example, we found that consuming a diet rich in saturated fat promotes a pro-inflammatory profile in the adipose tissue independent of weight gain, illustrating the impact that a small shift in the type of dietary fat has on health.
We also found that overweight insulin-sensitive subjects may benefit more from a high-nutrient-quality diet than insulin-resistant subjects for weight losss. These studies have greatly increased our understanding of why certain foods and diets might work for one individual but not for another individual, thereby establishing the basis for the concept of precision nutrition. In more recent studies, we additionally applied real-time sensors such as continuous glucose and physical activity monitors in human dietary intervention studies, thus paving the way for the next level in precision nutrition.
Determine endo- and exogenous factors and underlying mechanisms that can explain differences in response to meals and diets.
Glucose and triglyceride responses to meals vary from day to day and vary greatly between persons consuming the same meal. The goal is to identify both endo- and exogenous factors and mechanisms that determine differences in glucose and triglyceride responses to meals between and within persons. In the REBUS project, we have explored this goal by performing a completely controlled dietary intervention with repeated meals and 9 weeks of continuous glucose monitoring. The second approach is to identify endo- and exogenous factors and mechanisms that can explain long-term differences in responses to diet. In the Belly Fat study, we observed in an exploratory analysis that depending on the insulin-resistant state, people lost different amounts of weight on the same high-quality diet, thus pointing to the relevance of metabolic phenotype in the response to diet. In both approaches, we leverage nutrigenomic tools such as transcriptomics and metabolomics to identify potential mechanisms and subgroups of responders.
Identification of and precision nutrition in metabolic subgroups
Another research aim is to identify new metabolic subgroups from comprehensive phenotyping data of new or already completed studies. For example, within the “Immune Fitness” project, we are trying to identify subgroups of people with different states of immune fitness by comprehensive phenotyping of the immune cells, intending to identify potential pathways that underly the state of immune fitness and that can be targeted by diet. This feeds into my fourth goal, which is to use existing knowledge to identify and test the best diet for known metabolic subgroups, as was performed in the PERSON study. In this hugely challenging and complex study, we investigated two different diets that were hypothesized to be either optimal or suboptimal depending on the metabolic phenotype of a person.
Development of a digital twin that can give automatically generated personalized dietary advice that fits the person’s metabolic needs, values and preferences.
All these above mentioned studies provide a wealth of data that can subsequently be used to build computational simulation and prediction models using computational modelling and machine learning. They all provide answers that can be used as building blocks for a personalized smart healthcare tool, such as a digital twin. The ultimate aim is to develop a digital twin that can give automatically generated personalized dietary advice that not only fits the person’s metabolic needs, but also the persons’ values and preferences. There is an urgent call for such a tool as the worldwide increase in the development of overweight-related chronic metabolic diseases is paralleled by a steep rise in healthcare costs. Hence, this tool can not only improve personal health but can also reduce the pressure on the healthcare system. In the WUR-funded Flagship and digital twin project ‘Me, My Diet and I’ we explored the building of a digital twin that can give personalized dietary advice to reduce the personal after-meal triglyceride and glucose response.
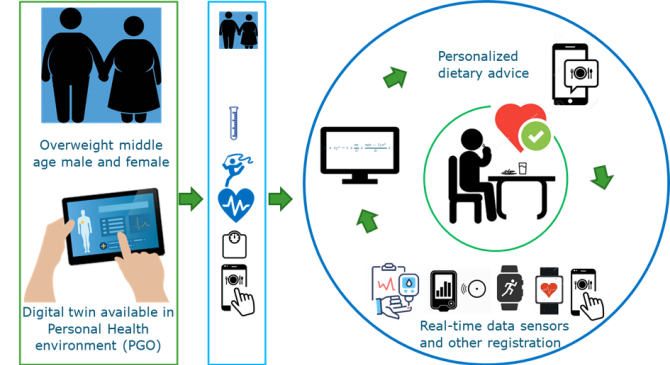
Figure of the anticipated digital twin