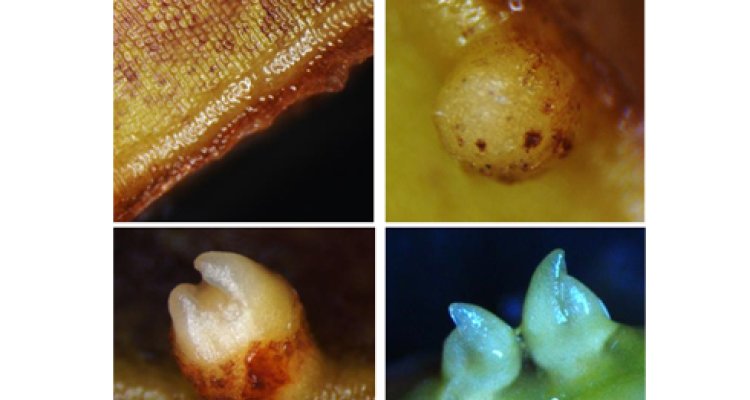
Effect of chromatin remodelling chemicals on bulblet regeneration in Lily
Lilies belong to the botanical family Liliaceae, and possess a bulb as the main storage organ. A bulb contains specialized scales that serve as the primary storage tissue and the scales are basically modified leaves attached to a modified short stem. Lilies are artificially propagated at low cost through detached bulb-scales (procedure called scaling), and the scales can be placed entire or cut in pieces (explants). Once the scales/explants have been placed in the appropriate growing conditions (100% humidity and 20-25°C), the first apparent morphological change appear after 15-20 days of culture, and it is given by a thickening of the bottom part of the explant. Thereafter few “bumps” appear and will gradually develop into bulblets.
Bulblet regeneration depends on the cultivar and the position of the explant in the bulb-scale (basal vs apical). Such differences are reflected in the number of regenerated bulblets and kinetics of the process. We have noticed that apical explants of cultivar Rialto stop at the formation of “bumps” and are not able to develop further into bulblets.
A recent study indicated that Trichostatin A (TSA), an inhibitor of Histone De-acetylases HDACs, strongly promotes bud growth in wild-type rice (Yoshida et al., 2012). When active, HDACs induce chromatin condensation, causing gene transcriptional repression. Hence, treating cultured explants with TSA may avoid any stop in bulblet regeneration and therefore may promote bulblet regeneration in lily.
Our approach
The aim of this experiment is to test whether TSA can influence bulblet regeneration capacity and speed of the process in lily bulb-scales.
Methodology
- Apical and basal explants of three cultivars differing in regeneration capacity will be cultured through scaling. During culturing, molecular analyses will be performed to determine exact developmental stages and to unravel the processes targeted by the chemical treatment. More specifically, the following activities are planned:Standardizing the time and concentration of TSA treatments.
- Staging explants twice a week for a period of at least 6 weeks.
- RNA isolation from regenerated bulblets at different time-points.
- Detailed morphological characterization (phenotyping).
- Search, selection and primer design of candidate genes that serve as markers to follow the regeneration process at the molecular level and tocorrelate acetylation levels.
- Quantification of gene expression for marker genes through qRT-PCR.
- Measurement of global acetylation levels and for selected target loci.
Techniques
Tissue culture, microscopy, expression studies (qRT-PCR), RNA/DNA isolation, DNA sequencing, bioinformatics (identification of marker genes, BLAST, etc.).