Single Domain Antibodies (VHHs)
Single domain antibody fragments (sdAbs) from camelids retain full antigen-binding capacity. These so-called VHHs or nanobodies offer advantages compared to conventional antibodies. They possess unique properties such as small size, excellent solubility, superior stability, quick clearance from blood and deep tissue penetration. These distinctive qualities make them useful tools in a diverse array of applications such as biosensing, affinity-capture, protein crystallisation, molecular imaging, tools for diagnosis and therapeutics.
What VHHs services do we offer?
Wageningen Bioveterinary Research (WBVR) offers the following single domain antibody services:
- Looking for VHHs for your research? We have an elaborate collection of VHHs for various antigens. The majority of them are directed against veterinary infectious diseases.
- In need of a custom produced VHH/nanobody? We can generate single domain antibodies against antigens supplied by our customers and customise according to their requirement.
What technology do we use to generate VHHs?
There are many ways to generate single domain antibodies in vivo and in vitro. The most common is generating an immunised or naive library, and screening by phage/yeast display. The most successfully applied platform technology in the context of antibody discovery is phage display. With the controlled selection and screening strategies, display technologies allow the generation of single domain antibodies against the defined antigens, their conformations or epitopes.
What are the project phases?
We offer you VHH isolation services against a wide variety of antigens, including native antigens, toxins and self-antigens. We have developed a naive
VHH phage display library from 47 animals (11 camels, 13 alpacas and
23 llamas) with a size of 4.4 × 109, along with our immunised
libraries to cater your requirements. Usually, we conduct two to four rounds of panning followed by screening of individual clones by ELISA to obtain high affinity binders.

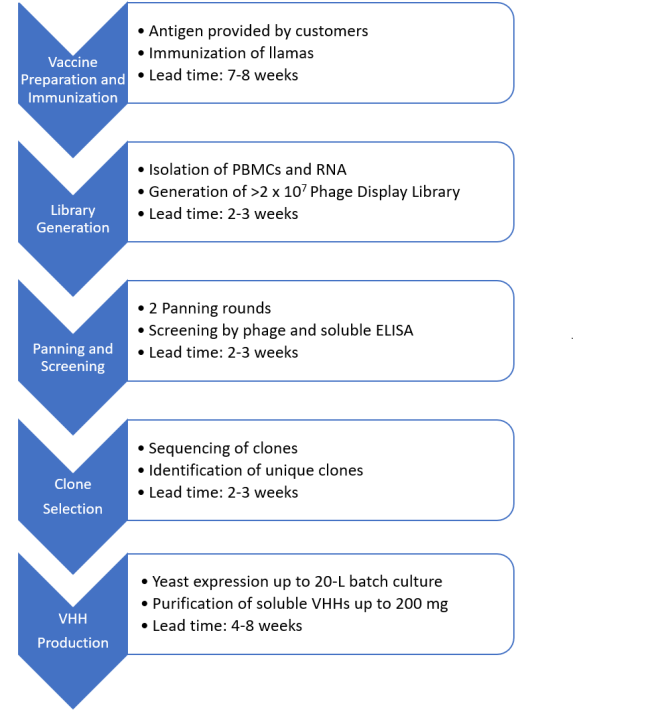
1. Immune phage display library
2. Immune phage display library, along with screening
We will immunise the llamas against the antigen provided by customers. Our deliverables will be unique clone sequences after screening the phage display library generated from the immunised llamas in 15-17 weeks, or the purified VHHs from the selected clones in 20-22 weeks.
Flow chart: Immune phage display library and deep sequencing
3. Naive library screening
See our research showcase about our naive VHH library project:
We have generated a naive VHH phage display library from 21 llamas and 2 camels. Our deliverables will be unique clone sequences selected from the naive llama or camel libraries in 5-6 weeks, or the purified VHHs from the selected clones in 4-8 weeks.
Flow chart: Naive library screening

4. Customized project
5. Affinity measurement
6. Deep sequencing
Which VHHs do we offer?
We offer many single domain antibodies from llamas, developed at our institute over the last three decades. These VHHs from llamas are produced in baker’s yeast and sold at 100 microgram to several mg quantities in unlabelled and biotinylated form. Especially a unique panel of foot-and-mouth disease virus (FMDV) and influenza A virus (IAV) binding VHHs are available. These are excellent reagents for quality control of vaccines. See our list of references below for more information on available VHHs.
View our key references
-
Small-Scale Secretory VHH Expression in Saccharomyces cerevisiae
In: Single-Domain Antibodies - Springer - ISBN: 9781071620755 - p. 159-179. -
Structural and molecular basis for foot-and-mouth disease virus neutralization by two potent protective antibodies
Protein & Cell (2022), Volume: 13, Issue: 6 - ISSN 1674-8018 - p. 446-453. -
A novel single-domain antibody multimer that potently neutralizes tetanus neurotoxin
Vaccine: X (2021), Volume: 8 - ISSN 2590-1362 -
Novel capsid-specific single-domain antibodies with broad foot-and-mouth disease strain recognition reveal differences in antigenicity of virions, empty capsids, and virus-like particles
Vaccines (2021), Volume: 9, Issue: 6 - ISSN 2076-393X -
Multimeric single-domain antibody complexes protect against bunyavirus infections
eLife (2020), Volume: 9 - ISSN 2050-084X -
Isolation of single-domain antibody fragments that preferentially detect intact (146s) particles of foot-and-mouth disease virus for use in vaccine quality control
Frontiers in Immunology (2017), Volume: 8 - ISSN 1664-3224 -
Effect of thiomersal on dissociation of intact (146S) foot-and-mouth disease virions into 12S particles as assessed by novel ELISAs specific for either 146S or 12S particles
Vaccine (2011), Volume: 29, Issue: 15 - ISSN 0264-410X - p. 2682-2690. -
Properties, production and applications of camelid single-domain antibody fragments
Applied Microbiology and Biotechnology (2007), Volume: 77, Issue: 1 - ISSN 0175-7598 - p. 13-22. -
Selection and optimization of proteolytically stable llama single-domain antibody fragments for oral immunotherapy
Applied Microbiology and Biotechnology (2006), Volume: 72, Issue: 3 - ISSN 0175-7598 - p. 544-551.
More information about our team
| Conny van Solt-Smits | Llama immunisation, blood sampling, processing and RNA isolation |
| Marga van Hagen-van Setten | Llama immunisation and VHH production in yeast and purification |
| Sandra van de Water | Library generation and screening of libraries |
| Nishi Gupta | Library generation, screening of libraries and selection of clones, antibody engineering |
| Michael Harmsen | Project leader |

