Project
Proteins and food structure
Protein Glass Transitions

- Financial support: FOM IPP "Bio(Related)Materials"
- PhD Student: Jacob Bouman
- collaboration with Paul Venema, Food Physics, Wageningen University

These protein/solvent dynamical transitions, or “protein glass transitions” have various consequences at the meso-(nanometer...micrometer) and macro (>micrometer) scale. The aim of this proposal is to link the conditions where various degrees of freedom of proteins and solvent freeze out, to the consequences at the meso- and macro scale and to finally obtain a full “state diagram” for selected globular proteins, using a combination of physical chemical techniques: spectroscopy, calorimetry, mechanical testing, microscopy, etc.
Enzymatic protein-cross linking

- Financial support: FOM IPP "Bio(Related)Materials"
- PhD Students: Yunus Saricay, Surender Dhayal
- collaboration with Peter Wierenga, Food Chemistry, Wageningen University
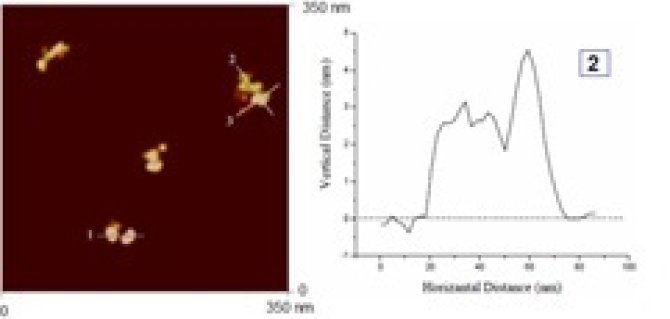
Enzymatic crosslinking of proteins is used in food structuring to modify the functional properties of proteins, but there is very little understanding of the relation between the structure of the cross-linked proteins and the resulting changes in the functional properties, With the aim of ultimately allowing for controlled enzymatic crosslinking to give protein products with controlled functionality (in both food and non-food application), we are studying the process of enzymatic protein crosslinking at colloidal lengthscales using on-line scattering, on-line spectroscopy as well as AFM and EM. Also, using well-characterized crosslinked protein products, we investigate the relation between the architecture and size of the cross-linked protein products, and their functional properties: heat stability, rheology, foaming, etc.
Dense protein particles for high protein foods

- Financial support: TI Food and Nutrition, project B1002 "High Protein Foods"
- PhD student: Dilek saglam
- collaboration with Paul Venema, Food Physics, Wageningen University
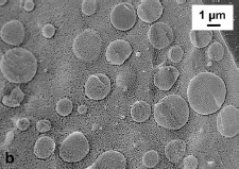

Nutrition studies have shown that high protein diets can be important in combatting obesitas and are beneficial for special groups such as the elderly. A general problem in formulating such foods is that they are sensitive to extensive protein aggregation in processing steps such as pasteurization and UHT that involve heating. How can we separate protein content from protein structuring in such formulations ? We are looking at the possibility to put away all the protein in inert dense protein particles, leaving sufficient continuous phase to allow for various forms of structuring. The research involves both developing procedures to prepare such particles, and studies on the behaviour of such particles in mixed systems.
Sağlam, D.;Venema, P.;de Vries, R.;Sagis, L.;van der Linden, E"Preparation of high protein micro-particles using two-step emulsification"
Food Hydrocolloids 25, 1139-1148 (2011)http://dx.doi.org/10.1016/j.foodhyd.2010.10.011