Persbericht
Aangepaste bacterie zet aardolie rechtstreeks om in bouwstenen voor kunststoffen
Een bacterie heeft een langgekoesterde wens van chemici in vervulling laten gaan. De met speciale enzymen uitgeruste E. coli bacterie maakt bouwstenen voor kunststoffen zoals polyesters rechtstreeks uit alkanen, een aardoliecomponent, zonder veel energie te verbruiken. Dat ontdekte onderzoeker Youri van Nuland in een studie waarop hij 20 oktober aan Wageningen University & Research promoveert. Een patent is aangevraagd.
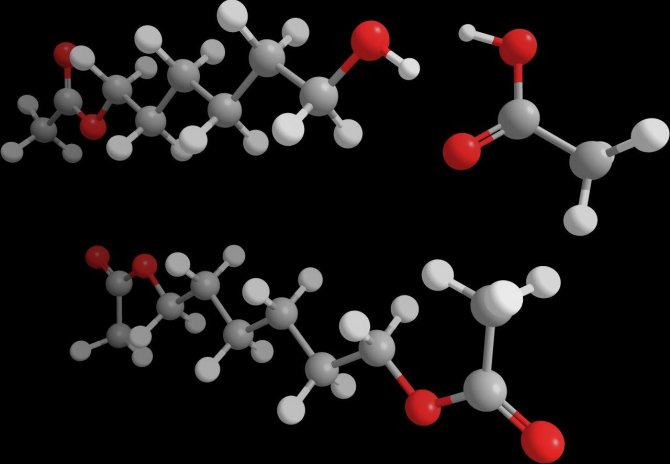
Om kunststoffen te maken moeten de bouwstenen aan elkaar gekoppeld worden. Elke bouwsteen heeft voor de koppeling aan de uiteinden twee speciale chemische groepen nodig, vergelijkbaar met haakjes of oogjes (figuur 1). Belangrijke bouwstenen zijn de zgn. alkaandiolen. Dat zijn alkanen, zoals propaan of butaan, met twee alcoholgroepen (‘haakjes’) aan de uiteinden. Ze kunnen gebruikt worden voor de productie van polyesters, polyurethanen, polyamides en andere kunststoffen.
De alkaandiol-bouwstenen die nu op de markt zijn worden vaak gemaakt uit de complexere grondstoffen acetyleen of benzeen, via een aantal energie-intensieve stappen waarbij grote hoeveelheden van de broeikasgassen kooldioxide en lachgas vrijkomen. Per kilogram product is de emissie zes kilogram CO2. Per jaar bedraagt de productie van butaandiol zo’n 1,8 miljoen ton.
Chemisch verlanglijstje
Het ligt daarom voor de hand om deze alkaandiol-bouwstenen rechtstreeks uit de eenvoudige en ruim voorradige alkanen te maken, door beide uiteinden van deze moleculen rechtstreeks te voorzien van een alcoholgroep, een ‘haakje’. Deze omzetting kost weinig energie en levert geringe hoeveelheden broeikasgassen op. Het realiseren van deze omzetting staat daarom sinds het begin van de petrochemie hoog op het verlanglijstje.
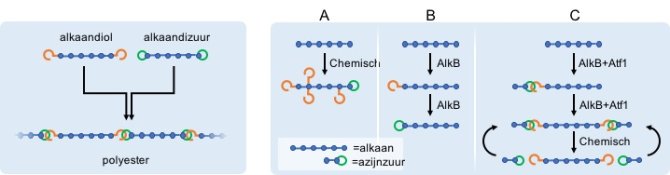
Toch lukte het vele industriële en universitaire laboratoria tot nu toe niet om deze directe route voor de synthese van bouwblokken voor de kunststoffabricage te realiseren. Het bleek dat tijdens het proces niet alleen de buitenste koolstofatomen van de alkanen werden voorzien van een alcoholgroep, maar dat dat ook aan de binnenste koolstofatomen zo’n ‘haakje’ werd aangeplakt. De chemische reactie was dus niet specifiek genoeg en leverde zo ongewenste bijproducten op. Bovendien werden de alcoholgroepen weer verder omgezet in zuurgroepen (figuur 2A), of werd het hele molecuul verbrand tot kooldioxide en water.
Enzymen
Het enzym alkaanhydroxylase (AlkB) is echter wel in staat zeer specifiek alleen de buitenste koolstofatomen van alkanen van alcoholgroepen te voorzien. Maar ook dit enzym verandert de alcoholgroepen in zuurgroepen. Bovendien kunnen zij dit aan slechts één kant van het alkaanmolecuul (figuur 2B). Een patstelling in de wens van de chemici, zo leek het.
Doorbraak
Maar de promovendus bij de Wageningse groep Bioprocess Engineering, Youri van Nuland, is er nu in geslaagd om met het AlkB-enzym toch de gewenste omzetting van alkanen tot alkaandiolen te realiseren. Hij rustte via genetische modificatie een bacteriestam van E. coli uit met de enzymen AlkB en alcohol acetyltransferase (Atf1). Het Atf1-enzym stelt de door AlkB gevormde alcoholgroep snel veilig, door het met azijnzuur te laten reageren tot een stabiele ester voordat het verder kan worden omgezet in een zuurgroep. Verrassend daarbij is dat AlkB nu wel in staat is ook het andere eind van het alkaan te voorzien van een alcoholgroep en dat Atf1 dit ook weer tot ester laat reageren. De gevormde esters van een alkaandiol zijn gemakkelijk en zonder veel energieverbruik om te zetten naar het gewenste alkaandiol (figuur 2C).
Youri van Nuland toonde deze omzetting op laboratoriumschaal aan met alkanen variërend van butaan tot decaan. Verdere optimalisatie en schaalvergroting is nodig om er een industrieel proces van te maken. Op de vinding is een patent aangevraagd.