Nieuws
Publication: A DNA Binding Bromodomain Containing Protein Interacts with and Reduces Rx1-Mediated Immune Response to Potato Virus X
Plant innate immunity is mediated by a group of specialized immune receptors, a majority of which belongs to the class of Nucleotide-Binding Leucine-Rich Repeats (NB-LRR). NB-LRRs act interacellularly to detect effector molecules derived from the pathogen, triggering a set of potent defence response.
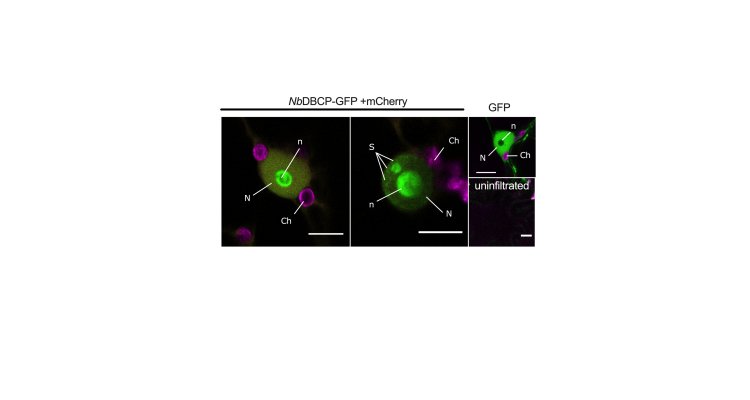
This typically includes dynamic transcriptional changes in the nucleus. In line with this, some NB-LRR proteins are known to recruit and directly modulate the activities of transcription factors. For example, the Rx1 immune receptor was previously shown to physically associate with and co-ordinate the DNA binding activity of the GLK1 transcription factor. Here, we show that Rx1 can co-exist with additional components in the nucleus. Based on a Yeast-2-Hybrid screening, a Bromodomain-containing protein (NbDBCP) was identified as an interactor of the Rx1-CC domain. In additional immunoprecipitation assays, we confirm that NbDBCP can likewise form complexes with full-length Rx1 in planta. Functional studies in the model plant Nicotiana Benthamiana demonstrate that NbDBCP suppresses Rx1-mediated defence against PVX and the pro-immune component GLK1. The former is dependent upon an intact bromodomain. Our findings collectively demonstrate that Rx1 can interact and co-ordinate the activities of nuclear-associated factors at the chromatin towards defence.