
Nieuws
Science: Improved degradation of micro pollutants in waste water
Waste water treatment plants are relatively ineffective to remove micro-pollutants such as diclofenac and metoprolol. Ph.D. researcher Wenbo Liu improved existing technologies to degrade these components using anaerobic conditions as well as bacteria.
During the last decades, waste water treatment technologies have improved dramatically. Removing organic substances and nutrients, like phosphorous and nitrogen, are highly effective. However, increasing amounts of pharmaceutical residues end up in waste water and are not sufficiently removed with conventional technologies. As a result, these substances end up in natural water systems. Despite their low concentrations, their toxicity pose an increasing threat to the ecosystem as well as the drinking water supply. For example, in The Netherlands diclofenac, a non-steroid anti-inflammatory drug (NSAID) (fig. 1a) and beta-blocker metoprolol (fig. 1b) is used by more than one million people annually.
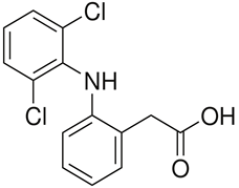
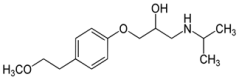
Often, as much as half of these compounds are not taken up by the body, but excreted in urine. In addition, breakdown products are excreted as well and may pose additional risks to the environment and affect our ecosystem and drinking water safety. With ageing populations, amounts of pharmaceuticals discharged into aquatic ecosystems will only increase. ‘Diclofenac is among the most persistent and toxic pharmaceuticals’, says ETE scientist Wenbo Liu. ‘It shows toxic effects already at low concentrations.’ Therefore, EU placed this pharmaceutical on the watch list of persistent compounds, requiring environmental monitoring. Nevertheless, release of pharmaceuticals into the environment is not yet regulated.
More efficient methods
‘Current methods to remove micro-pollutants from waste water include advanced oxidation, adsorption to activated carbon’, Liu explains. ‘These methods are often not very efficient. Therefore, we were looking for improved, more efficient methods to degrade recalcitrant pharmaceuticals, like diclofenac and metoprolol.’ By experimenting with different reaction conditions and the absence or presence of microorganisms to help degradation, Liu managed to improve the well-established manganese-oxide (MnO2) oxidation method. In the absence of oxygen (anoxic conditions), degradation of diclofenac was largely improved, he found out. After 24 hours of reaction, removal efficiencies were about 60% with oxygen, but increased to over 90% under conditions without oxygen, an improvement of 50% (fig.2).
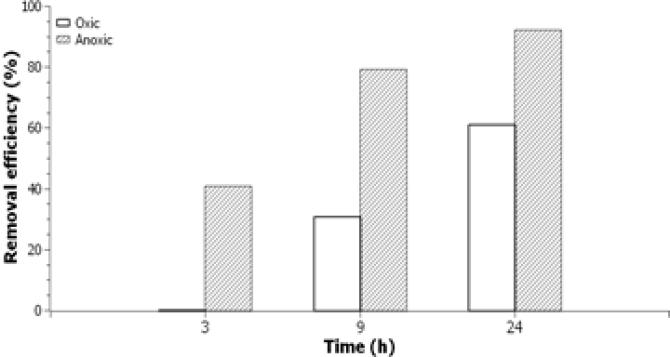
Hard to degrade
To enhance diclofenac degradation even more, Liu combined anaerobic MnO2 oxidation with additional breakdown by microorganisms. This didn’t result in a better removal efficiency though. ‘Based on other research, we expected to see a better degradation of diclofenac, because certain bacteria are able to break down persistent compounds’, says Liu. ‘But possibly diclofenac is too difficult to degrade for bacteria due to the presence of two stable phenyl rings in the molecule.’
Metoprolol, on the other hand, showed significant improved degradation when combining anaerobic MnO2 oxidation and microorganisms. After 50 days, using MnO2 oxidation only, metoprolol concentrations dropped from 11 mg/l to 7 mg/l, a decreased of about 36%. Including microorganisms spectacularly decreased concentrations to less than 1 mg/l.
