Preclinical and clinical studies
As an independent contract research organization (CRO), Wageningen Bioveterinary Research (WBVR) is well suited for preclinical and clinical trials for public and private clients.
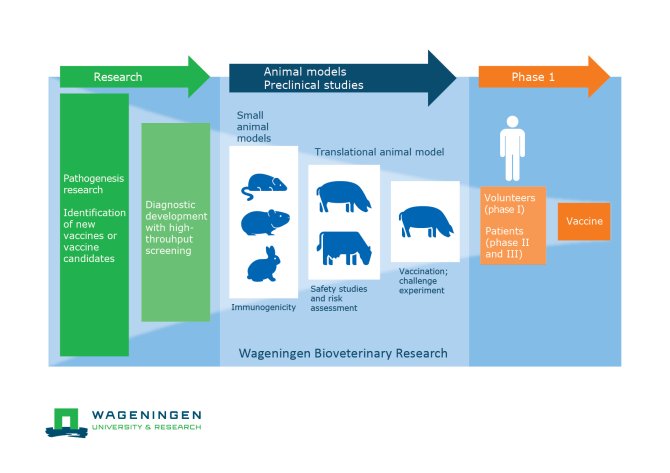
Using our versatile animal facilities, WBVR can perform trials for vaccine testing, safety studies as well as nutritional studies to test feed components. We can perform trials in healthy animals, but can also use a range of pathogen in animal models.
International standards and guidelines
Depending on the requirements of our clients, trials can be conducted following the Good Clinical Practical VICH guidelines, can be compliant with the European Food Safety Authority (EFSA), follow the European Pharmacopoeia or the guidelines of the European Medicines Evaluation Agency (EMEA). All of our extraneous agents, batch control and potency testing services are supervised by our QA officers.
Animal models for human health too
We can facilitate (pre-)clinical trials for veterinary and human pharmaceuticals as well as for feed producers. Our animal models either represent the target animals or can be translational models for human health. Our experts on epidemiology can advise on the most effective study design, including the required number of animals as well as on statistical methods for analyses. We can request ethical permission for animal experiments at the Dutch legislation institute on behalf of our clients.
Customized studies
In addition to performing animal trials, we can support additional analyses to be conducted in our laboratory facilities. Pathogen load can be determined using classical microbiological techniques or molecular biological techniques. Host responses can be monitored using different techniques like serology, immunology, and pathology. For all analyses, we can offer customized studies following the specific requirements and protocols of our clients.
Contact our experts
Please feel free to contact the experts of our contract research organization (CRO) if you have a question concerning preclinical or clinical studies.
Infographic: phases in studies