Project
Anaerobic ammonium removal and electricity recovery with bioelectrochemical system
Removal of nitrogen compounds from wastewater is essential to prevent pollution of receiving water bodies. In wastewater, nitrogen is mostly present in the form of ammonium. Conventional ammonium removal technologies are biological processes, including nitrification and denitrification. They are energy-intensive due to aeration and carbon resource input. Besides the energy consumption, another downside is the production of NOx. NOx is a greenhouse gas, which emission should be minimized. For these reasons, novel and sustainable ammonium elimination processes should be developed. Bioelectrochemical systems (BESs) have gained attention as an alternative to ammonium treatment. They have been demonstrated to have efficient and energy-efficient ammonium removal performance.
Technological challenge
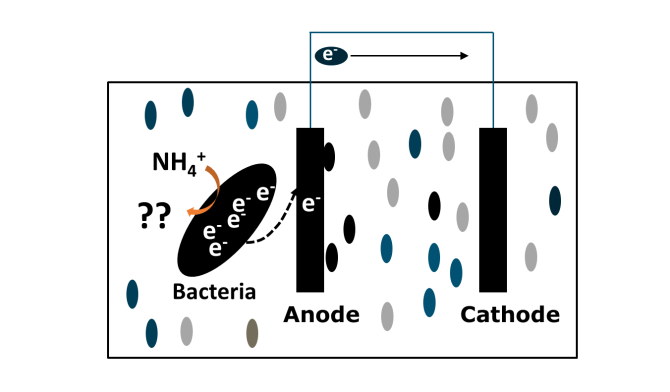
BESs utilize electrochemically active microorganism to catalyze the reaction in the electrode. Electrons driven by the oxidation of pollutants are transferred into the anode, resulting in pollutants removal. The electrons travel from anode to cathode offer the possibility of converting chemical energy into electrical energy.
In BESs, ammonium oxidation may occur under anaerobic conditions. In this project, we will explore how ammonium-oxidizing bacteria (AOB)/ nitrite-oxidizing bacteria (NOB) are able to directly transfer electrons to anode instead of oxygen and enable the recovery of electricity. Using bacteria to oxidize ammonium at the anode of a BES results in a reduced energy input for aeration, reduction in N2O emissions, and energy recovery from ammonium.
The technological challenge is to prove the concept of ammonium oxidation at the bio-anode and understanding the pathway of ammonium oxidation. Subsequently, we will explore the effect of operational conditions on the ammonium removal efficiency and electricity recovery. This study is expected to lay a fundamental basis for developing a more sustainable ammonium removal process using BESs.